Think about a time where you were injured, perhaps you hit your thumb with a hammer. 
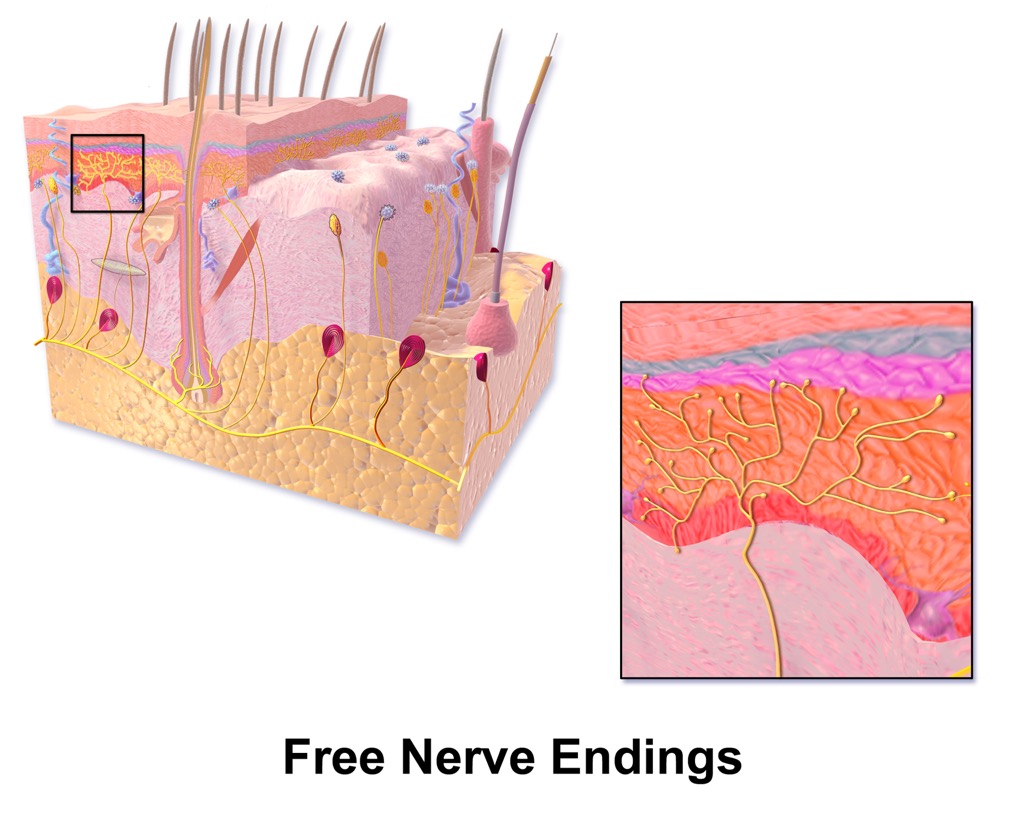
We can often perceive this difference in the form of “first pain” and “second pain”. The Aδ and C fibers transmit information to the CNS at different speeds because of their different myelination levels. ’Nociceptors’ by Casey Henley is licensed under a Creative Commons Attribution Non-Commercial Share-Alike (CC BY-NC-SA) 4.0 International License. Nociceptor fibers can either by unmyelinated (C fibers) or lightly myelinated (A delta fibers). Activation of these nociceptors lead to perceived pain sensation. Free nerve endings in the skin and other tissues respond to tissue damage. The C fibers carry upwards of 70% of noxious signals within the body. 
The unmyelinated C fibers have an even smaller diameter (~1 μm) and slower conduction velocity (~1 m/s) than the Aδ fibers. They have larger receptive fields, which results in a lower ability to locate pain. Type C fibers tend to be polymodal and are activated by a range of stimuli including mechanical, chemical, and thermal. Type Aδ II fibers have a low threshold for thermal stimulation and activate in extremely hot or extremely cold environments. Type Aδ I fibers have a low threshold for mechanical stimulation and activate following intense pressure or an incision on the skin. They are lightly myelinated, smaller in diameter (~5 μM), and transmit signals slower than the Aβ fibers that transmit touch information, around ~25 m/s. The Aδ fibers have small receptive fields which allows for precise location of pain. Nociceptors are located throughout the body in skin, muscles, and viscera, but there are no nociceptors in the CNS. Depending on the type of stimulus, either Aδ fibers or C fibers are activated to transmit information to the CNS. 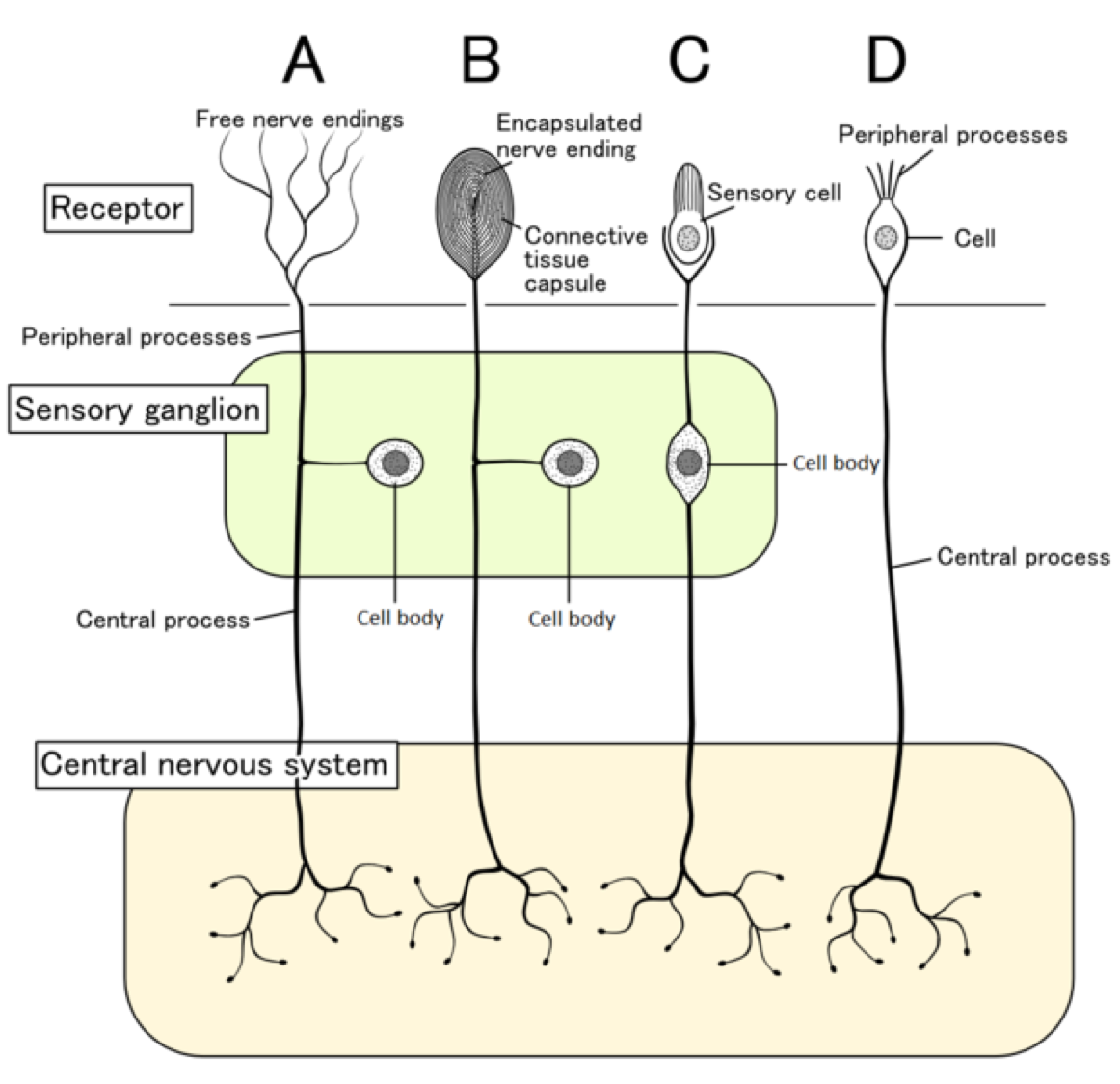
A mutation in this ion channel that prevents it from functioning can results in the inability to feel pain.Īctivation of nociceptors usually occurs in response to tissue damage or the threat of damage. A special type of sodium channel, Na1.7, is present only in nociceptor fibers. Lidocaine, a common local anesthetic, dulls pain by blocking these voltage-gated sodium channels, preventing signal transmission. Once depolarization occurs in response to a painful stimulus, action potential propagation depends on the action of voltage-gated sodium and potassium channels. When a nociceptor is activated, it causes the opening of mechanically-gated ion channels that allow for cation influx, resulting in cell depolarization. Similar to other somatosensory mechanoreceptors, the sensory end of the pseudounipolar neurons have their cell soma in the dorsal root ganglion. These free nerve endings are found closest to the surface of the skin when compared to other mechanoreceptors. Many pain-detecting neurons respond to more than one type of noxious stimulus and are called polymodal nociceptors. They are expressed on free nerve endings that innervate the skin. Nociceptors can detect a variety of noxious stimuli, ranging from crush (mechanical), acid (chemical), and high heat (thermal). Pain detection is carried out by nociceptors that are located in the periphery at the surface of the skin. When we feel pain, we are more likely to protect the injured site to prevent further damage, making the sensation of pain beneficial to our overall well-being. Detection and avoidance of pain is a highly-adaptive behavior that can improve the odds of survival chances of an animal. Nociception is a sensory process that provides signals that trigger pain when in the presence of a noxious stimulus. Our study greatly expands the molecular characterization of C-LTMRs and suggests that this particular population of neurons shares some molecular features with A beta and A delta low-threshold mechanoreceptors.Pain is a complex experience that has somatosensory, psychological, and affective components. We validated 48 MRGPD- and 68 C-LTMRs-enriched genes using a triple-staining approach, and the Ca(v)3.3 channel, found to be exclusively expressed in C-LTMRs, was validated using electrophysiology. Cluster analysis of RNA-seq profiles of the purified neuronal subsets revealed 486 and 549 genes differentially expressed in MRGPRD-expressing neurons and C-LTMRs, respectively. Here, we used a mouse model that genetically marks C-LTMRs and MRGPRD(+) neurons in combination with fluorescent cell surface labeling, flow cytometry, and RNA deep-sequencing technology (RNA-seq). The molecular mechanisms underlying these diametrically opposite functions are unknown. Cutaneous C-unmyelinated MRGPRD(+) free nerve endings and C-LTMRs innervating hair follicles convey two opposite aspects of touch sensation: a sensation of pain and a sensation of pleasant touch. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
